|
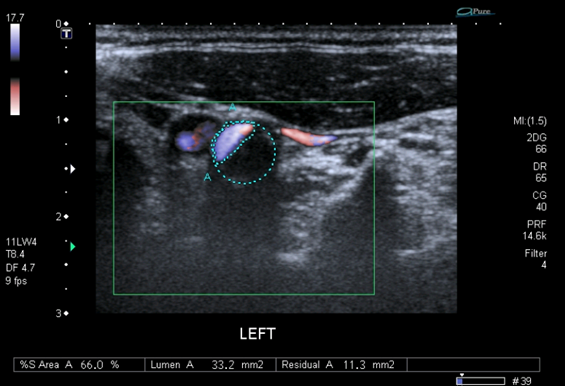
Under NCD 20.7, the TCAR Surveillance Project does not require the study sites to get approval from the Medicare Administrative Contractors. View the CMS coverage letters detailing approval of the TCAR Surveillance Project for high-risk and standard surgical risk patients. CMS will reimburse hospitals and physicians for symptomatic and asymptomatic patients at high risk or standard risk for traditional carotid artery surgery who participate in the TCAR Surveillance Project. According to the National Coverage Determination (NCD) 20.7 for Percutaneous Transluminal Angioplasty, the TCAR Surveillance Project is an FDA-approved post-approval carotid stenting study. TCAR Surveillance ProjectĮffective September 1, 2016, hospitals participating in the Society for Vascular Surgery (SVS) Vascular Quality Initiative (VQI) receive CMS coverage for Transcarotid Artery Revascularization (TCAR) procedures entered into the VQI Carotid Artery Stenting module. The degree of carotid artery stenosis shall be measured by duplex Doppler ultrasound or contrast tomography angiography (CTA) or magnetic resonance angiography (MRA) or carotid artery angiography and recorded in the patient’s medical records.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
